Summary: The Parkinson’s drug entacapone disrupts the gut microbiome by inducing iron deficiency, favoring the growth of potentially harmful bacteria like E. coli. Using advanced molecular techniques, researchers identified that entacapone interferes with iron availability, a critical resource for many gut microbes.
This mechanism of gut dysbiosis highlights broader implications for how human-targeted drugs impact microbial health. Mitigating these effects, such as by supplementing iron selectively to the gut, may help reduce side effects and support better treatment outcomes.
This work underscores the intricate relationship between drugs, the gut microbiome, and overall health. The findings pave the way for microbiome-conscious drug designs in the future.
Key Facts:
- Entacapone disrupts the gut microbiome by inducing iron deficiency, favoring bacteria like E. coli that thrive under low iron conditions.
- Advanced techniques, including heavy water labeling and Raman spectroscopy, revealed subtle microbial activity changes beyond abundance measurements.
- Ensuring targeted iron delivery to the gut could reduce drug-induced dysbiosis while maintaining therapeutic efficacy.
Source: University of Vienna
In a groundbreaking new study, conducted within the framework of the FWF-funded Cluster of Excellence “Microbiomes drive Planetary Health”, scientists from the University of Vienna, in collaboration with the University of Southampton, Aalborg University and Boston University, have revealed that the widely prescribed Parkinson’s disease drug entacapone significantly disrupts the human gut microbiome by inducing iron deficiency.
The study, published in Nature Microbiology, provides new insights into the often-overlooked impact of human-targeted drugs on the microbial communities that play a critical role in human health.
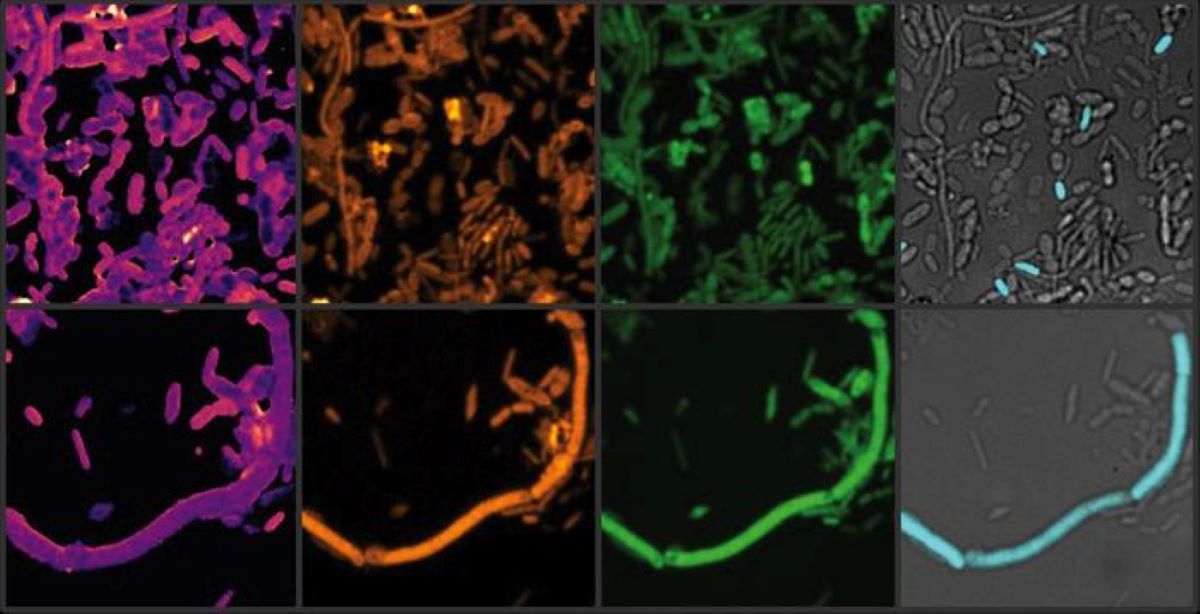
While it is well established that antibiotics can significantly disrupt the human gut microbiome, emerging research shows that a wide range of human-targeted drugs—particularly those used to treat neurological conditions—can also profoundly affect the microbial communities living in our bodies.
Despite their intended therapeutic effects on different organs, these drugs can inadvertently disrupt the balance of gut microbes, leading to potential health consequences.
Until now, most studies investigating these interactions relied either on patient cohort analyses affected by many confounding factors or on experiments using isolated gut bacteria, which do not fully capture the complexity of the human microbiome.
A Novel Study Design to Investigate Drug-Bug Interactions
Using a novel experimental approach, the international team studied the effects of two drugs—entacapone and loxapine, a medication for schizophrenia—on fecal samples from healthy human donors.
They incubated the samples with therapeutic concentrations of these drugs, then analyzed the impact on the microbial communities using advanced molecular and imaging techniques, including heavy water labeling combined with Stimulated Raman Spectroscopy (SRS).
The team discovered that loxapine and even more so entacapone severely inhibited many microbiome members, while E. coli dramatically expanded in the presence of entacapone.
“The results were even more striking when we examined microbial activity, rather than just their abundance,” explained Fatima Pereira, lead author of the study and former Postdoctoral researcher at the University of Vienna.
“The heavy water-SRS method allowed us to observe the subtle yet significant changes in the gut microbiome, which are often missed in traditional abundance-based measurements.”
Entacapone Induces Iron Starvation, Favors Pathogenic Microbes
The researchers hypothesized that entacapone might interfere with iron availability in the gut, a crucial resource for many microbes. Their experiments confirmed that adding iron to fecal samples containing entacapone counteracted the drug’s microbiome-altering effects.
Further investigation revealed that E. coli, which thrived under these conditions, carried a highly efficient iron-uptake system (enterobactin siderophore). This system allowed the bacteria to overcome iron starvation and proliferate, even in the presence of the drug.
“By showing that entacapone induces iron deficiency, we have uncovered a new mechanism of drug-induced gut dysbiosis, in which the drug selects for E. coli and other potentially pathogenic microbes well adapted to iron limiting conditions,” said Michael Wagner, scientific director of the Excellence Cluster and vice-head of the Centre for Microbiology and Environmental Systems Science (CeMESS) at the University of Vienna.
Wider Implications for Drug-Microbiome Interactions
This discovery has broader implications for understanding how other human-targeted drugs might affect the gut microbiome. Several drugs, including entacapone, contain metal-binding catechol groups, suggesting that this mechanism could be a more common pathway for drug-induced microbiome alterations.
The findings also present an opportunity to mitigate the side effects of drugs like entacapone. By ensuring sufficient iron availability to the large intestine, it may be possible to reduce dysbiosis and the gastrointestinal issues that often accompany Parkinson’s disease treatment.
“The next step is to explore how we can modify drug treatments to better support the gut microbiome,” said Wagner.
“We are looking at strategies to selectively deliver iron to the large intestine, where it can benefit the microbiome without interfering with drug absorption in the small intestine.”
About the Study: This study was conducted as part of the FWF-funded Cluster of Excellence “Microbiomes drive Planetary Health,” a collaborative research initiative including eight leading Austrian Research Institutions.
About this neuropharmacology and microbiome research news
Author: Alexandra Frey
Source: University of Vienna
Contact: Alexandra Frey – University of Vienna
Image: The image is credited Xiaowei Ge (Boston University)
Original Research: Open access.
“The Parkinson’s disease drug entacapone disrupts gut microbiome homeostasis via iron sequestration” by Michael Wagner et al. Nature Microbiology
Abstract
The Parkinson’s disease drug entacapone disrupts gut microbiome homeostasis via iron sequestration
Many human-targeted drugs alter the gut microbiome, leading to implications for host health. However, the mechanisms underlying these effects are not well known.
Here we combined quantitative microbiome profiling, long-read metagenomics, stable isotope probing and single-cell chemical imaging to investigate the impact of two widely prescribed drugs on the gut microbiome.
Physiologically relevant concentrations of entacapone, a treatment for Parkinson’s disease, or loxapine succinate, used to treat schizophrenia, were incubated ex vivo with human faecal samples.
Both drugs significantly impact microbial activity, more so than microbial abundance. Mechanistically, entacapone can complex and deplete available iron resulting in gut microbiome composition and function changes. Microbial growth can be rescued by replenishing levels of microbiota-accessible iron.
Further, entacapone-induced iron starvation selected for iron-scavenging gut microbiome members encoding antimicrobial resistance and virulence genes.
These findings reveal the impact of two under-investigated drugs on whole microbiomes and identify metal sequestration as a mechanism of drug-induced microbiome disturbance.
This was shown first on: https://neurosciencenews.com/parkinsons-neuropharmacology-microbiome-28105/


